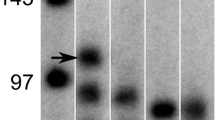
Summary
To catalog factors that may contribute to the completion of myogenesis, we have been looking for molecular differences between BC3H1 and C2C12 cells. Cells of the BC3H1 tumor line, though myogenic, are nonfusing, and withdraw from the cell cycle only reversibly, whereas cells of the C2C12 line fuse, differentiate terminally, and express several muscle-specific gene products that BC3H1 cells do not. Relative to C2C12 cells, BC3H1 cells underaccumulated cyclin-dependent kinase inhibitor p21 and underaccumulated transcripts for p21, GADD45, CDO, decorin, osteopontin, H19, fibronectin, and thrombospondin-1 (tsp-1). Levels of accumulation of H19, tsp-1, and larger isoforms of fibronectin messenger ribonucleic acid (mRNA) were found to increase in response to expression of myogenic regulatory factors as shown by their accumulation in differentiated myogenically converted 10T1/2 cells but not in 10T1/2 fibroblasts. BC3H1s accumulated a temperature-insensitive, geldanamycin-sensitive, misfolded form of p53 incapable of transactivating a p53 responsive reporter, consistent with underexpression of p21, GADD45, and tsp-1. BC3H1 and C2C12 cells were similar with respect to upregulation of p27 protein, downregulation of mitogen-activated protein kinase phosphatase-1 (MKP-1) protein, upregulation of retinoblastoma (Rb) mRNA, and nuclear localization of hypophosphorylated Rb. Cells of both lines expressed the muscle-specific 1b isoform of MEF2D. Although nonfusing in the short term, after more than 18 d in differentiation medium, some cultures of BC3H1 cells formed viable multinucleated cells in which the nuclei did not reinitiate synthesis of DNA in response to serum. Our findings suggest participation of tsp-1 and specific isoforms of fibronectin in myogenesis and suggest additional avenues of research in myogenesis and oncogenesis.
Similar content being viewed by others
References
Adams, J.; Lawler, J. Cell-type specific adhesive interactions of skeletal myoblasts with thrombospondin-1. Mol. Biol. Cell 5:423–437; 1994.
Andres, V.; Walsh, K. Myogenin expression, cell cycle withdrawal, and phenotypic differentiation are temporally separable events that precede cell fusion upon myogenesis. J. Cell Biol. 132:657–666; 1996.
Bennett, A. M.; Tonks, N. K. Regulation of distinct stages of skeletal muscle differentiation by mitogen-activated protein kinases. Science 278:1288–1291; 1997.
Bernards, R.; Shackleford, G. M.; Gerber, M. R., et al. Structure and expression of the murine retinoblastoma gene and characterization of its encoded protein. Proc. Natl. Acad. Sci. USA 86:6474–6478; 1989.
Blau, H.; Chiu, C.; Wester, C. Cytoplasmic activation of human nuclear genes in stable heterokaryons. Cell 32:1171–1180; 1983.
Block, N. E.; Miller, J. B. Expression of MRF4, a myogenic helix-loop-helix protein, produces multiple changes in the myogenic program of BC3H-1 cells. Mol. Cell. Biol. 12:2484–2492; 1992.
Boulter, J.; Patrick, J. Purification of an acetylcholine receptor from a nonfusing muscle cell line. Biochemistry 16:4900–4908; 1977.
Bradford, M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Anal. Biochem. 72:248–254; 1976.
Brandan, E.; Fuentes, M. D.; Andrade, W. The proteoglycan decorin is synthesized and secreted by differentiated myotubes. Eur. J. Cell Biol. 55:209–216; 1991.
Braun, T.; Bober, E.; Buschhausen-Denker, G.; Kohtz, S.; Grzeschik, K. H.; Arnold, H. H.; Kohtz, S. Differential expression of myogenic determination genes in muscle cells: possible autoactivation by the Myf gene products. EMBO J. 8:3617–3625; 1989.
Brennan, T.; Edmondson, D.; Olson, E. Aberrant regulation of MyoD contibutes to the partially defective myogenic phenotype of BC3H1 cells. J. Cell Biol. 110:929–937; 1990.
Burnette, W. Western blotting: electrophoretic transfer of proteins from SDS-polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radiocoordinated protein A. Anal. Biochem. 112:195–203; 1981.
Chen, P.; Scully, P.; Shew, J.; Wang, J. Y. J.; Lee, W. Phosphorylation of the retinoblastoma gene product is modulated during the cell cycle and cellular differention. Cell 58:1193–1198; 1989.
Chomczynski, P.; Sacchi, N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 162:156–159; 1987.
Coppola, J. A.; Lewis, B. A.; Cole, M. D. Increased retinoblastoma gene expression is associated with late stages of differentiation in many different cell types. Oncogene 5:1731–1733; 1990.
Dameron, K. M.; Volpert, O. V.; Tainsky, M. A.; Bouck, N. Control of angiogenesis in fibroblasts by p53 regulation of thrombospondin-I. Science 265:1582–1584; 1994.
Dasgupta, G.; Momand, J. Geldanamycin prevents nuclear translocation of mutant p53. Exp. Cell Res. 237:29–37; 1997.
Davis, R. L.; Weintraub, H.; Lassar, A. B. Expression of a single transfected cDNA converts fibroblasts to myoblasts. Cell 51:987–1000; 1987.
Diatchenko, L.; Lukyanov, S.; Lau, Y. F.; Siebert, P. D. Suppression subtractive hybridization: a versatile method for identifying differentially expressed genes. Methods Enzymol. 303:349–380; 1999.
el-Deiry, W. S.; Tokino, T.; Velculescu, V. E., et al. WAF1, a potential mediator of p53 tumor suppression. Cell 75:817–825; 1993.
Ellis, L.; Clauser, E.; Morgan, D. O.; Edery, M.; Roth, R. A.; Rutter, W. J. Replacement of insulin receptor tyrosine residues 1162 and 1163 compromises insulin-stimulated kinase activity and uptake of 2-deoxyglucose. Cell 45:721–732; 1986.
Feinberg, A. P.; Vogelstein, B. Addendum: a technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal. Biochem. 137:266; 1984.
Finlay, C. A.; Hinds, P. W.; Tan, T.-H.; Eliyahu, D; Oren, M.; Levine, A. J. Activating mutations for transformation by p53 produce a gene product that forms an hsc70-p53 complex with an altered half-life. Mol. Cell. Biol. 8:531–539; 1988.
Friend, S. p53: a glimpse at the puppet behind the shadow play. Science 265:334–335; 1994.
Gannon, J. V.; Greaves, R.; Iggo, R.; Lane, D. P. Activating mutations in p53 produce a common conformational effect. A monoclonal antibody specific for the mutant form. EMBO 9:1595–1602; 1990.
Gorman, C. M.; Moffat, L. F.; Howard, B. H. Recombinant genomes which express chloramphenicol acetyltransferase in mammalian cells. Mol. Cell. Biol. 2:1044–1051; 1982.
Gossett, L. A.; Kelvin, D. J.; Sternberg, E. A.; Olson, E. N. A new myocyte-specific enhancer-binding factor that recognizes a conserved element associated with multiple muscle-specific genes. Mol. Cell. Biol. 9:5022–5033; 1989.
Graham, F.; Van der Eb, A. A new technique for the assay of infectivity of human adenovirus 5 DNA. Virology 52:456–467; 1973.
Gu, W.; Schneider, J. W.; Condorelli, G.; Kaushal, S.; Mahdavi, V.; Nadal-Ginard, B. Interaction of myogenic factors and the retinoblastoma protein mediates muscle cell commitment and differentiation. Cell 72:309–324; 1993.
Guo, K.; Wang, J.; Andres, V.; Smith, R.; Walsh, K. MyoD-induced expression of p21 inhibits cyclin-dependent kinase activity upon myocyte terminal differentiation. Mol. Cell. Biol. 15:3823–3829; 1995.
Halevy, O.; Novitch, B. G.; Spicer, D. B.; Skapek, S. X.; Rhee, J.; Hannon, G. J.; Beach, D.; Lassar, A. Correlation of terminal cell cycle arrest of skeletal muscle with induction of p21 by MyoD. Science 267:1018–1021; 1995.
Hall, C. V.; Jacob, P. E.; Ringold, G. M.; Lee, F. Expression and regulation of Escherichia coli lacZ gene fusions in mammalian cells. J. Mol. Appl. Genet. 2:101–109; 1983.
Han, J.; Jiang, Y.; Li, Z.; Kravchenko, V. V.; Ulevitch, R. J. Activation of the transcription factor MEF2C by the MAP kinase p38 in inflammation. Nature 386:296–299; 1997.
Hantai, D.; Rao, J. S.; Reddy, B. R.; Festoff, B. W. Developmental appearance of thrombospondin in neonatal mouse skeletal muscle. Eur. J. Cell Biol. 55:286–294; 1991.
Harlow, E.; Crawford, L. V.; Pim, D. C.; Williamson, N. M. Monoclonal antibodies specific for simian virus 40 tumor antigen. J. Virol. 39:861–869; 1981.
Harlow, E.; Lane, D. ed. Antibodies—a laboratory manual. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press; 1988:447.
Haupt, Y.; Maya, R.; Kazaz, A.; Oren, M. Mdm2 promotes the rapid degradation of p53. Nature 387:296–299; 1997.
Huppi, K.; Siwarski, D.; Dosik, J., et al. Molecular cloning, sequencing, chromosomal localization and expression of mouse p21 (Wafl). Oncogene 9:3017–3020; 1994.
Jiang, Z.; Liang, P.; Leng, R., et al. E2F1 and p53 are dispensable, whereas p21 Wafl/Cip1 cooperates with Rb to restrict endoreduplication and apptosis during skeletal myogenesis. Dev. Biol. 227:28–41; 2000.
Jiminez, B.; Volpert, O. V.; Crawford, S. E.; Febbraio, M.; Silverstein, R. L.; Bouck, N. Signals leading to apoptosis-dependent inhibition of neovascularization by thrombospondin-1. Nat Med. 6:41–48; 2000.
Kang, J.-S.; Gao, M.; Feinleib, J.; Cotter, P.; Guadagno, S.; Krauss, R. S. CDO: an oncogene-, serum-, and anchorage-regulated member of the Ig/Fibronectin Type III repeat family. J. Cell Biol. 138:203–213; 1997.
Kang, J.-S.; Mulieri, P. J.; Hu, Y.; Taliana, L.; Krauss, R. S. BOC, an Ig superfamily member associates with CDO to positively regulate myogenic differentiation. EMBO J. 21:114–124; 2002.
Kang, J.-S.; Mulieri, P.; Miller, C.; Sassoon, D.; Krauss, R. S. CDO, a roborelated cell surface protein that mediates myogenic differentiation. J. Cell Biol. 143:403–413; 1998.
Kastan, M. B.; Zhan, Z.; el-Deiry, W. S., et al. A mammalian cell cycle checkpoint pathway utilizing p53 and GADD45 is defective in ataxiatelangiectasia. Cell 71:587–597; 1992.
Kubbutat, M.; Jones, S.; Vousden, K. Regulation of p53 stability by Mdm2. Nature 387:299–303; 1997.
Kurschat, P.; Mauch, C. Mechanisms of metastasis. Clin. Exp. Dermatol. 25:482–489; 2000.
Mal, A.; Chattopadhyay, D.; Ghosh, M.; Poon, R.; Hunter, T.; Harter, M. p21 and retinoblastoma protein control the absence of DNA replication in terminally differentiated muscle cells. J. Cell Biol. 149:281–292; 2000.
Martelli, F.; Cenciarelli, C.; Santarelli, G.; Polikar, B.; Felsani, A.; Caruso, M. MyoD induces retinoblastoma gene expression during myogenic differentiation. Oncogene 9:3579–3590; 1994.
Martin, J. F.; Miano, J. M.; Hustad, C. M.; Copeland, N. G.; Jenkins, N. A.; Olson, E. N.: A Mef2 gene that generates a muscle-specific isoform via alternative mRNA splicing. Mol. Cell. Biol. 14:1647–1656; 1994.
Melo, F.; Carey, D.; Brandan, E. Extracellular matrix is required for skeletal muscle differentiation but not myogenin expression. J. Cell. Biochem. 62:227–239; 1996.
Miller, J. B. Myogenic programs of mouse muscle cell lines: expression of myosin heavy chain isoforms, MyoD1, and myogenin. J. Cell Biol. 111:1149–1159; 1990.
Milligan, L.; Antoine, D.; Bisbal, C.; Weber, M.; Brunel, C.; Forne, T.; Cathala, G. H19 gene expression is up-regulated exclusively by stabilization of the RNA during muscle cell differentiation. Oncogene 19:5810–5816; 2000.
Milner, J.; Medcalf, E. A. Cotranslation of activated mutant p53 with wild type drives the wild-type p53 protein into the mutant conformation. Cell 65:765–774; 1991.
Miner, J. H.; Wold, B. Herculin, a fourth member of the MyoD family of myogenic regulatory genes. Proc. Natl. Acad. Sci. USA 87:1989–1093; 1990.
Miner, J. H.; Wold, B. J. c-myc inhibition of myoD and myogenin-initiated myogenic differentiation. Mol. Cell. Biol. 11:2842–2851; 1991.
Mittnacht, S.; Weinberg, R. A. G1/S phosphorylation of the retinoblastoma protein is associated with an altered affinity for the nuclear compartment. Cell 65:381–393; 1991.
Nishimura, T.; Futami, E.; Taneichi, A.; Mori, T.; Hattori, A. Decorin expression during development of bovine skeletal muscle and its role in morphogenesis of the intramuscular connective tissue. Cells Tissues Organs 171:199–214; 2002.
Novitch, B.; Mulligan, G.; Jacks, T.; Lassar, A. Skeletal muscle cells lacking the retinoblastoma protein display defects in muscle gene expression and accumulate in S and G2 phases of the cell cycle. J. Cell Biol. 135:441–456; 1996.
Novitch, B.; Spicer, D.; Kim, P.; Cheung, W.; Lassar, A. pRb is required for MEF2-dependent gene expression as well as cell-cycle arrest during skeletal muscle differentiation. Curr. Biol. 20:449–459; 1999.
Olson, E.; Sternbgerg, E.; Hu, J.; Spizz, G.; Wilcox, C. Regulation of myogenic differentiation by type β transforming growth factor. J. Cell Biol. 103:1799–1805; 1986.
Pachnis, V.; Belayew, A.; Tilghman, S. Locus unlinked to α-fetoprotein under the control of the murine raf and Rif genes. Proc. Natl. Acad. Sci. USA 81:5523–5527; 1984.
Pandey, S.; Wang, E. Cells en route to apoptosis are characterized by the upregulation of c-fos, c-myc, c-jun, cdc2, and Rb phosphorylation, resenbling events of early cell-cycle traverse. J. Cell. Biochem. 58:135–150; 1995.
Park, D. J.; Chumakov, A. M.; Miller, C. W.; Pham, E. Y.; Koeffler, H. P. P53 transactivation through various p53-responsive elements. Mol. Carcinog. 16:101–108; 1996.
Parker, S.; Eichele, G.; Zhang, P.; Rawls, A.; Bradley, A.; Olson, E.; Harper, J.; Elledge, S. p53-independent expression of p21Cipl in muscle and other terminally differentiating cells. Science 267:1024–1027; 1995.
Pinter, A.; Chen, T.; Lowry, A.; Cortez, N.; Silagi, S. Ecotropic murine leukemia virus-induced fusion of murine cells. J. Virol. 57:1048–1054; 1986.
Polyak, K.; Lee, M.; Edijument-Bromage, H.; Koff, A.; Roberts, J. M.; Tempst, P.; Massague, J. Cloning of p27KIPI, a cyclin-dependent kinase inhibitor and a potential mediator of extracellular antimitogenesis signals. Cell 78:59–66; 1994.
Porrello, A.; Cerone, M.; Coen, S., et al. p53 regulates myogenesis by triggering the differentiation activity of pRb. J. Cell Biol. 151:1295–1303; 2000.
Ragheb, J. A.; Anderson, W. F. pH-independent murine leukemia virus ecotropic envelope-mediated cell fusion: implication for the role of the R peptide and p12E TM in viral entry. J. Virol. 68:3220–3231; 1994.
Rein, A.; Mirro, J.; Haynes, J. G.; Ernst, S. M.; Nagashima, K. Function of the cytoplasmic domain of a retroviral transmembrane protein: p15E–p2E cleavage activates the membrane fusion capability of the murine leukemia virus Env protein. J. Virol. 68:1773–1781; 1994.
Reznikoff, C. A.; Brankow, D. W.; Heidelberger, C. Establishment and characterization of a cloned line of C3H mouse embryo cells sensitive to postconfluence inhibition of division. Cancer Res. 33:3231–3238; 1973.
Rhodes, S. J.; Konieczny, S. F. Identification of MRF4: a new member of the muscle regulatory factor gene family. Genes Dev. 3:2050–2061; 1989.
Riquelme, C.; Larrain, J.; Schonherr, E.; Henriquez, J.; Kresse, H.; Brandan, E. Antisense inhibition of decorin expression in myoblasts decreases cell responsiveness to transforming growth factor beta and accelerates skeletal muscle differentiation. J. Biol. Chem. 276:3589–3596; 2001.
Rosenthal, S.; Hsiao, D.; Silverman, L. An insulin-like growth factor-II (Igf-II) analog with highly selective affinity for Igf-II receptors stimulates differentiation, but not Igf-I receptor down-regulation in muscle cells. Endocrinology 134:38–44; 1994.
Santra, M.; Mann, D.; Mercer, E.; Skorski, T.; Calabretta, B.; Iozzo, R. Ectopic expression of decorin protein core causes a generalized growth suppression in neoplastic cells of various histogenetic origin and requires endogenous p21, an inhibitor of cyclin-dependent kinases. J. Clin. Invest. 100:149–157; 1997.
Schneider, J. W.; Gu, W.; Zhu, L.; Mahdavi, V.; Nadal-Ginard, B. Reversal of terminal differentiation mediated by p107 in Rb−/− muscle cells. Science 264:1467–1471; 1994.
Schubert, D.; Harris, A.; Devine, C.; Heineman, S. Characterization of a unique muscle cell line. J. Cell Biol. 61:398–413; 1974.
Schwarzbauer, J. E.; Patel, R. S.; Fonda, D.; Hynes, R. O. Multiple sites of alternative splicing of the rat fibronectin gene transcript. EMBO J. 6:2573–2580; 1987.
Sharp, S. B.; Kim, S.; Lee, M., et al. The levels of vascular smooth as well as skeletal muscle actin mRNAs differ substantially among both myoblast and fibroblast lines with different skeletal myogeic potentials. In Vitro Cell. Dev. Biol. 31A:749–751; 1995.
Sharp, S. B.; Villalvazo, M.; Espinosa, A.; Damle, S.; Padilla, X.; Gonzalez, R.; Vu, S. BC3H1 myogenic cells produce an infectious ecotropic murine leukemia virus. In Vitro Cell. Dev. Biol. 38:378–381; 2002.
Sherr, C. Cancer cell cycles. Science 274:1672–1677; 1996.
Soddu, S.; Blandino, G.; Skardigli, R., et al. Interference with p53 protein inhibits hematopoetic and muscle differentiation. J. Cell Biol. 134:193–204; 1996.
Sodek, J.; Ganss, B.; McKee, J. Osteopontin. Crit. Rev. Oral Biol. Med. 11:279–303; 2000.
Southern, P. J.; Berg, P. Transformation of mammalian cells to antibiotic resistance with a bacterial gene under control of the SV40 early region promoter. J. Mol. Appl. Genet. 1:327–341; 1982.
Steenstrup, T.; Hannon, K. Isolation of a spontaneously fusing BC3H1 muscle cell line: fusion alters the response to serum stimulation. In Vitro Cell. Dev. Biol. 36A:241–248; 2000.
Strauch, A. R.; Reeser, J. Sequential expression of smooth muscle and sarcomeric alpha-actin isoforms during BC3H1 cell differentiation. J. Biol. Chem. 264:8345–8355; 1989.
Taubman, M. B.; Smith, C. W. J.; Izumo, S.; Grant, J. W.; Endo, T.; Andreadis, A.; Nadal-Ginard, B. The expression of sarcomeric muscle-specific contractile protein genes in BC3H1 cells: BC3H1 cells resemble skeletal myoblasts that are defective for commitment to terminal differentiation. J. Cell Biol. 108:1799–1806; 1989.
Thorburn, A. M.; Walton, P.; Feramisco, J. R. MyoD induced cell cycle arrest is associated with increased nuclear affinity of the Rb protein. Mol. Biol. Cell. 4:705–713; 1993.
Tollefsen, S.; Sadow, J.; Rotwein, P. Coordinate expression of insulin-like growth factor II and its receptor during muscle differentiation. Proc. Natl. Acad. Sci. USA 86:1543–1547; 1989.
Whitesell, L.; Sutphin, P. D.; Pulcini, E. J.; Martinez, J. D.; Cook, P. H. The physical association of multiple molecular chaperone proteins with mutant p53 is altered by geldanamycin, an hsp90-binding agent. Mol. Cell. Biol. 18:1517–1524; 1998.
Wilkin, F.; Paquette, J.; Ledru, E.; Hamelin, C.; Pollak, M.; Deal, C.; Mamelin, C. H19 sense and antisense transgenes modify insulin-like growth factor-II mRNA levels. Eur. J. Biochem. 267:4020–4027; 2000.
Yaffe, D.; Saxel, O. A myogenic cell line with altered serum requirements for differentiation. Differentiation 7:159–166; 1977.
Zabludoff, S.; Csete, M.; Wagner, R.; Yu, X.; Wold, B. J. p27 Kipl is expressed transiently in myotomes and enhances myogenesis. Cell Growth Differ. 9:1–11; 1998.
Zetser, A.; Gredinger, E.; Bengal, E. p38 mitogen-activated protein kinase pathway promotes skeletal muscle differentiation. Participation of the MEF2C transcription factor. J. Biol. Chem. 274:5193–5100; 1999.
Zhang, J.-M.; Zhao, X.; Wei, Q.; Paterson, B. M. Direct inhibition of G1 cdk kinase activity by MyoD promotes myoblast cell cycle withdrawal and terminal differentiation. EMBO J. 18:6983–6993; 1999.
Zhao, M.; Liguo, N.; Kravchenko, V., et al. Regulation of the MEF2 family of transcription factors by p38. Mol. Cell. Biol. 19:21–30; 1999.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sharp, S.B., Villalvazo, M., Huang, M. et al. Further characterization of BC3H1 myogenic cells reveals lack of P53 activity and underexpression of several P53 regulated and extracellular matrix-associated gene products. In Vitro Cell.Dev.Biol.-Animal 38, 382–393 (2002). https://doi.org/10.1290/1071-2690(2002)038<0382:FCOBMC>2.0.CO;2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/1071-2690(2002)038<0382:FCOBMC>2.0.CO;2